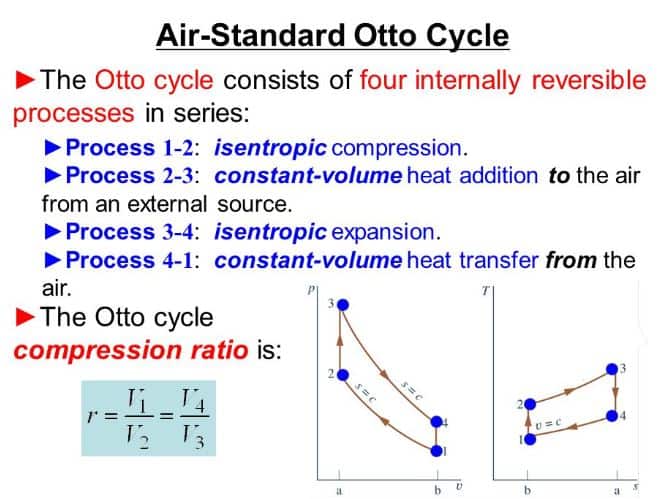
可用性和第二法|Thermal Interview Question and Answers
可用性和第二法|Thermal Interview ,viva ,oral Question and Answers
1.定义Clausius语句。
在循环过程中工作的自动机器是不可能在较低温度下在较低温度下在较低温度下将热量从身体转移到较高的外部机构。
2. What is Perpetual motion machine of the second kind?
A heat engine, which converts whole of the heat energy into mechanical work is known as Perpetual motion machine of the second kind.
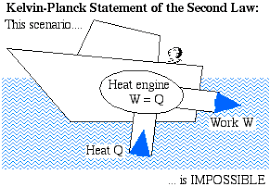
3.定义Kelvin Planck语句。
如果在单个固定温度下从单个储存器交换热量,则无法在完全循环中构造热力发动机以在完整的循环中产生网络。
4. Define Heat pump.
A heat pump is a device, which is working in a cycle and transfers heat from lower temperature to higher temperature.
5. Define Heat engine.
Heat engine is a machine, which is used to convert the heat energy into mechanical work in a cyclic process.
6. A heat engine with a thermal efficiency of 45 percent rejects 500 kJ/kg of heat. How much heat does it receive?
热效率=(QS-QR)/ QS
0.45 =(QS-500)/ QS
qs = 909 kj / kg
7.什么是逆转的热力发动机?
The reversed heat engine works on the principle of reversed Carnot cycle. The heat engine produces work by absorbing heat from source and liberating some heat to sink. The reversed heat engine transfers the heat from sink to the source with the help of external work.
8.热力发动机的假设是什么?
1.源和水槽保持在恒定温度。
2. The source and sink has infinite heat capacity.
9. State Carnot theorem.
It states that no heat engine operating in a cycle between two constant temperature heat reservoir can be more efficient than a reversible engine operating between the same reservoir.
10. What is meant by reversible process?
可逆过程是一种,它以这样的方式执行,即在过程的结论中,系统和周围环境都可以恢复到它们的初始状态,而不会产生宇宙剩余部分的任何变化。
11. What is meant by irreversible process?
两种物质和燃烧的混合也导致不可逆性。所有自发过程都是不可逆转的。
12. Explain entropy?
它是物质的重要热力学性质。它是分子障碍的衡量标准。它由S表示。通过接收或被拒绝绝对温度的热量获得可逆过程的熵变化的测量。
13.什么是绝对熵?
针对绝对零温度下的所有完美结晶固体测量的熵称为绝对熵。
14.当系统绝热时,可以说什么是系统中物质的熵变化?
Entropy change of the substance in a adiabatic system is always constant. (S=C) S1=S2
15. Define availability.
The maximum useful work obtained during a process in which the final condition of the system is the same as that of the surrounding is called availability of the system.
16.定义可用的能量和不可用的能量。
Available energy is the maximum thermal useful work under ideal condition. The remaining part, which cannot be converted into work, is known as unavailable energy.
17. What is a thermal energy reservoir? Explain the term source and sink.
A thermal reservoir, a short-form of thermal energy reservoir, or thermal bath is a thermodynamic system with a heat capacity that is large enough that when it is in thermal contact with another system of interest or its environment, its temperature remains effectively constant.
Source is a thermal reservoir, which supplies heat to the system and sink is a thermal reservoir, which takes the heat from the system.
18. What do you understand by the entropy principle?
The entropy of an isolated system can never decrease. It always increases and remains constant only when the process is reversible. This is known as principle of increase in entropy or entropy principle.
19. What are the important characteristics of entropy?
1.如果向系统提供热量,则熵会增加。
2. If the heat is rejected to the system then the entropy will decrease.
3. The entropy is constant for all adiabatic frictionless process.
4.如果在节流过程中,如果在没有工作的情况下降低了热量,则熵增加。
5. If the entropy is maximum, then there is a minimum availability for conversion in to work.
6. If the entropy is minimum then there is a maximum availability for conversion into work.
20.什么是逆转的Carnot热力发动机?Carnot Cycle的局限性是什么?
1. No friction is considered for moving parts of the engine.
2.不应该有任何热量损失。
21.定义概要过程。
概要过程也称为可逆的绝热过程。它是一种遵循PVY = C的法律的过程被称为熵过程。在此过程中,熵保持恒定,没有热量进入或离开气体。
22. Explain the throttling process.
When a gas or vapour expands and flows through an aperture of small size, the process is called as throttling process.
23. What are the Corollaries of Carnot theorem?
(i)在整个可逆发动机在具有固定温度的两个给定的热贮存器之间运行,具有相同的效率。
(ii)在两个储存器之间操作的任何可逆热发动机的效率与工作流体的性质无关,并且仅取决于储存器的温度。
24.定义 - 第二种PMM。
第二种运动机从单个储存器中连续地加热,并将其转换为等同的工作。因此它提供了100%的效率。
25.热泵和冰箱之间有什么区别?
Heat pump is a device which operating in cyclic process, maintains the temperature of a hot body at a temperature higher than the temperature of surroundings.
A refrigerator is a device which operating in a cyclic process, maintains the temperature of a cold body at a temperature lower than the temperature of the surroundings.
26. Define the term COP?
系数的性能is defined as the ratio of heat extracted or rejected to work input.
COP =热提取或拒绝/工作输入
28. Why Carnot cycle cannot be realized in practical?
(i) In a Carnot cycle all the four process are reversible but in actual practice there is no process irreversible.
(ii) There are two processes to be carried out during compression and expansion. For isothermal process the piston moves very slowly and for adiabatic process the piston moves as fast as possible. This speed variation during the same stroke of the piston is not possible.
(iii) It is not possible to avoid friction moving parts completely.
29. Why a heat engine cannot have 100% efficiency?
对于所有热发动机,系统和周围环境之间将存在热量损失。因此,我们无法将所有热量转换为有用的工作。
30. What are the processes involved in Carnot cycle.
Carnot cycle consist of
i) Reversible isothermal compression
ii)势尺寸压缩
iii)可逆等温膨胀
iv)等熵扩张
31. What are the causes of irreversibility?
Four of the most common causes of irreversibility are friction, unrestrained expansion of a fluid, heat transfer through a finite temperature difference, and mixing of two different substances.
32.国家热力学II法律陈述陈述。
Clausius声明状态“不可能在没有任何外力的循环过程中工作的自动机器,以在较高温度下将来自体内的热量从身体转移到体内。它考虑了两个热藏之间的热量的转变。